Quantum redactiones paginae "Anandamidum" differant
Content deleted Content added
pag. inst. |
mNo edit summary |
||
| Linea 4: | Linea 4: | ||
! colspan="2" | Cognitores |
! colspan="2" | Cognitores |
||
|- |
|- |
||
| style="font-size: 85%" | ''[[ChemSpider]]'' || style="font-size: 85%" | [http://www.chemspider.com/Chemical-Structure. |
| style="font-size: 85%" | ''[[ChemSpider]]'' || style="font-size: 85%" | [http://www.chemspider.com/Chemical-Structure.4445241.html 4445241] |
||
|- |
|- |
||
| style="font-size: 85%" | [[PubChem]] || style="font-size: 85%" | [https://pubchem.ncbi.nlm.nih.gov/compound/5281969 5281969] |
| style="font-size: 85%" | [[PubChem]] || style="font-size: 85%" | [https://pubchem.ncbi.nlm.nih.gov/compound/5281969 5281969] |
||
| Linea 21: | Linea 21: | ||
Effectus anandamidi cum praesentia [[receptorium cannabinoideum|receptoriorum cannabinoideorum]] nexi sunt. Putatur quoque anandamido effectus in [[morbus mentis|morbis mentis]] esse<ref>{{cite journal |authors=Gaetani S., Dipasquale P., Romano A., Righetti L., Cassano T., Piomelli D., Cuomo V. |title=The endocannabinoid system as a target for novel anxiolytic and antidepressant drugs |journal=International review of neurobiology |year=2009 |volume=85 |pages=57-72 |url=https://www.ncbi.nlm.nih.gov/pubmed/19607961}}</ref>. |
Effectus anandamidi cum praesentia [[receptorium cannabinoideum|receptoriorum cannabinoideorum]] nexi sunt. Putatur quoque anandamido effectus in [[morbus mentis|morbis mentis]] esse<ref>{{cite journal |authors=Gaetani S., Dipasquale P., Romano A., Righetti L., Cassano T., Piomelli D., Cuomo V. |title=The endocannabinoid system as a target for novel anxiolytic and antidepressant drugs |journal=International review of neurobiology |year=2009 |volume=85 |pages=57-72 |url=https://www.ncbi.nlm.nih.gov/pubmed/19607961}}</ref>. |
||
== Natura chemica == |
|||
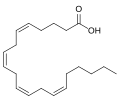
Compositum est anandamidum longa ex molecula [[Acidum arachidonicum|acidi arachidonici]] per amino secundario cum [[ethanol|aaethanolo]] (aeathanolamino) coniunctum. Nomen [[IUPAC]] est (5Z,8Z,11Z,14Z)-''N''-(2-hydroxy-aethyl)-icosa-5,8,11,14-tetraenamidum. |
|||
<gallery> |
|||
Fasciculus:Arachidonic acid.svg|[[Acidum arachidonicum]] |
|||
Fasciculus:Secondary-amine-2D-general.svg|Aminum secundarium |
|||
Fasciculus:Ethanolamine-2D-skeletal-B.png|Aethanolaminum |
|||
Fasciculus:Ethanol-2D-skeletal.svg|[[ethanol|Aethanolum]]<br />([[alcohol primarium]]) |
|||
Fasciculus:Primary-amine-2D-general.svg|Aminum primarium |
|||
</gallery> |
|||
== Notae == |
== Notae == |
||
Emendatio ex 08:38, 18 Ianuarii 2020
| Cognitores | |
|---|---|
| ChemSpider | 4445241 |
| PubChem | 5281969 |
| Natura chemica | |
| |

| |
| Formula summarum | C 22H 37NO 2 |
| Massa molaris | 347.53 g/mol |
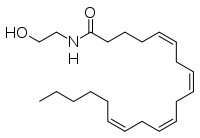
Anandamidum vel N-arachidonoylaethanolaminum (AEA) est neurotransmissor et unctoacidum ab acido arachidonico, uno unctoacidorum omega-6 essentialium, derivatum. Pars nominis prima de Lingua Sanscrita (ananda = felicitas) Lingua Sanscrita.
Effectus anandamidi cum praesentia receptoriorum cannabinoideorum nexi sunt. Putatur quoque anandamido effectus in morbis mentis esse[1].
Natura chemica
Compositum est anandamidum longa ex molecula acidi arachidonici per amino secundario cum aaethanolo (aeathanolamino) coniunctum. Nomen IUPAC est (5Z,8Z,11Z,14Z)-N-(2-hydroxy-aethyl)-icosa-5,8,11,14-tetraenamidum.
-
Aminum secundarium
-
Aethanolaminum
-
Aminum primarium
Notae
- ↑ Gaetani S., Dipasquale P., Romano A., Righetti L., Cassano T., Piomelli D., Cuomo V. (2009). "The endocannabinoid system as a target for novel anxiolytic and antidepressant drugs". International review of neurobiology 85: 57-72
Bibliographia
- Mechoulam R., Fride E.: The unpaved road to the endogenous brain cannabinoid ligands, the anandamides in “Cannabinoid Receptors” (ed. R. Pertwee), Academic Press, London. Pp. 233-258 (1995).