Fasciculus:Sodium chloride crystal.png
Appearance
Sodium_chloride_crystal.png (410 × 372 elementa imaginalia, magnitudo fasciculi: 52 chiliocteti, typus MIME: image/png)
Historia fasciculi
Presso die vel tempore fasciculum videbis, sicut tunc temporis apparuit.
| Dies/Tempus | Minutio | Dimensiones | Usor | Sententia | |
|---|---|---|---|---|---|
| recentissima | 23:27, 9 Februarii 2023 |  | 410 × 372 (52 chiliocteti) | Materialscientist | transparent background |
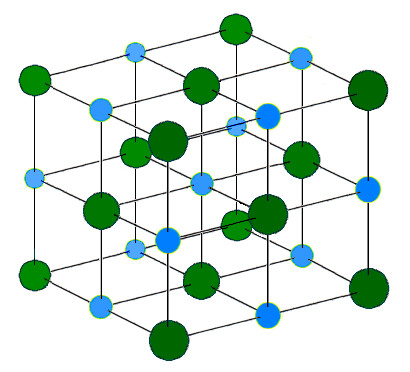
| 23:54, 14 Iunii 2005 |  | 410 × 372 (37 chiliocteti) | H Padleckas | The Sodium Chloride Crystal Structure. Each atom has six nearest neighbors, with octahedral geometry. This arrangement is known as ''cubic close packed'' (ccp). Light blue = Na<sup>+</sup> Dark green = Cl<sup>-</sup> |
Nexus ad fasciculum
Ad hunc fasciculum nectit:
Usus fasciculi per inceptus Vicimediorum
Quae incepta Vici fasciculo utuntur:
- Usus in am.wikipedia.org
- Usus in ar.wikipedia.org
- Usus in bs.wikipedia.org
- Usus in ca.wikipedia.org
- Usus in co.wikipedia.org
- Usus in cs.wikipedia.org
- Usus in de.wikipedia.org
- Usus in de.wikibooks.org
- Usus in el.wikipedia.org
- Usus in el.wikibooks.org
- Usus in en.wikipedia.org
- Usus in en.wikibooks.org
- Organic Chemistry/Foundational concepts of organic chemistry/Bonding
- General Chemistry/Introduction
- General Chemistry/Ionic bonding
- Organic Chemistry/Print version
- Adventist Youth Honors Answer Book/Nature/Rocks & Minerals - Advanced
- General Chemistry/Print version
- User:Jplego/Collections
- User:Jplego/Gen Chem
- Usus in en.wikiversity.org
- Usus in eo.wikipedia.org
- Usus in es.wikipedia.org
- Usus in fi.wikipedia.org
- Usus in fr.wikipedia.org
- Usus in fr.wikiversity.org
- Usus in fr.wiktionary.org
- Usus in ga.wikipedia.org
- Usus in he.wikipedia.org
- Usus in hr.wikipedia.org
- Usus in hu.wikipedia.org
- Usus in id.wikibooks.org
- Usus in it.wikipedia.org
- Usus in ko.wikipedia.org
- Usus in lt.wikipedia.org
- Usus in mk.wikipedia.org
View more global usage of this file.